
Under extremely high pressure, such as is found at the core of Jupiter, hydrogen does become metallic and behaves like an alkali metal see metallic hydrogen. Binary compounds of hydride with the alkali metals and some transition metals have been prepared. As in the halogens, only one additional electron is required to fill in the outermost shell of the hydrogen atom, so hydrogen can in some circumstances behave like a halogen, forming the negative hydride ion. Removal of its single electron requires considerably more energy than removal of the outer electron for the alkali metals. Hydrogen, with a solitary electron, is usually placed at the top of Group 1 of the periodic table, but it is not considered an alkali metal rather it exists naturally as a diatomic gas.
These elements all have one electron in their outermost shell, so the energetically preferred state of achieving a filled electron shell is to lose one electron to form a singly charged positive ion, or cation. The alkali metals are silver-colored (cesium has a golden tinge), soft, low- density metals, which react readily with halogens to form ionic salts, and with water to form strongly alkaline (basic) hydroxides. Potassium and rubidium possess a weak radioactive characteristic (harmless) due to the presence of long duration radioactive isotopes. They also tarnish easily and have low melting points and densities. As a result, in the laboratory they are stored under mineral oil.
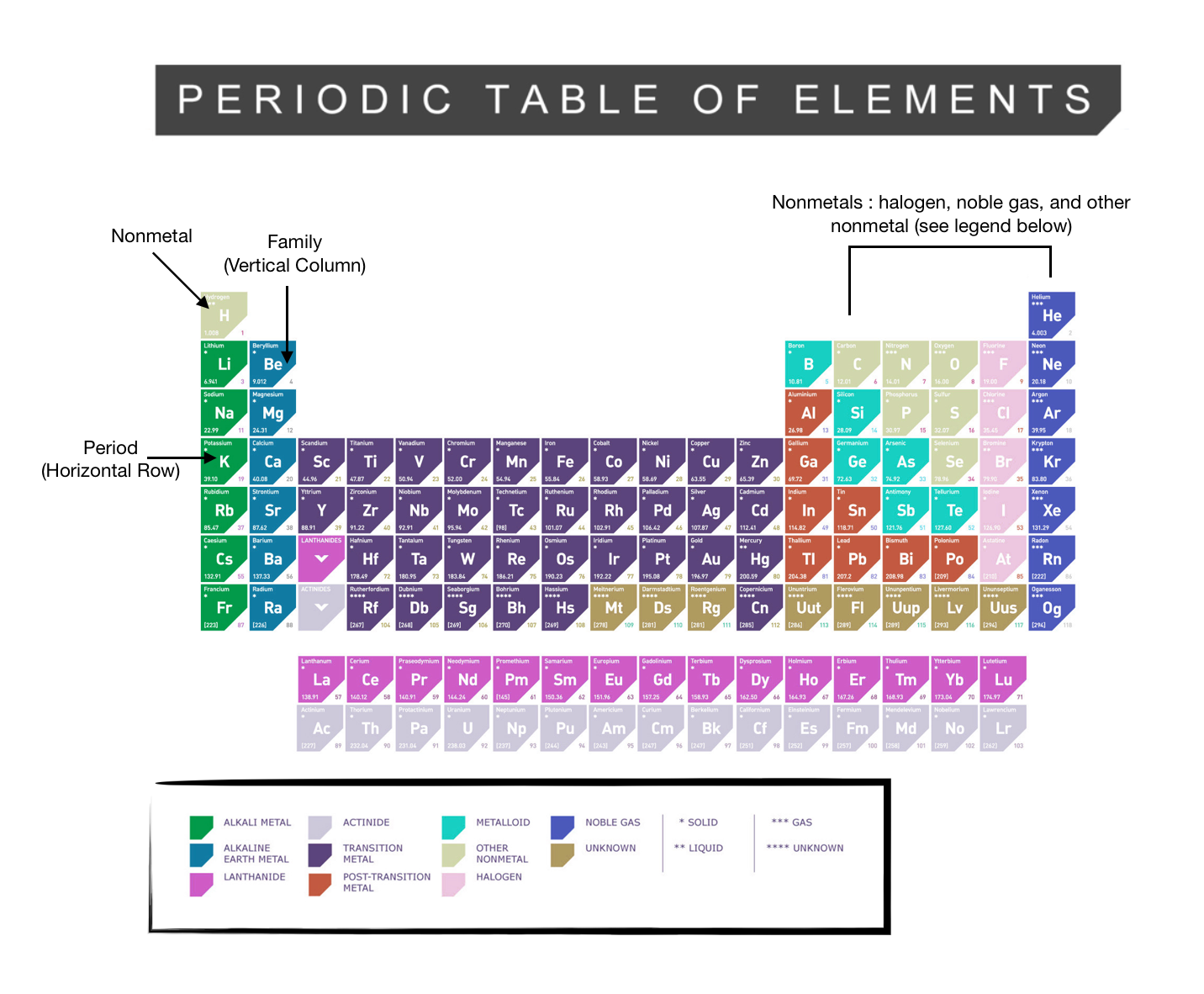
The alkali metals are all highly reactive and are rarely found in elemental form in nature. The alkali metals provide one of the best examples of group trends in properties in the periodic table, with well characterized homologous behavior down the group. (Note that hydrogen, although nominally also a member of Group 1, very rarely exhibits behavior comparable to the alkali metals).
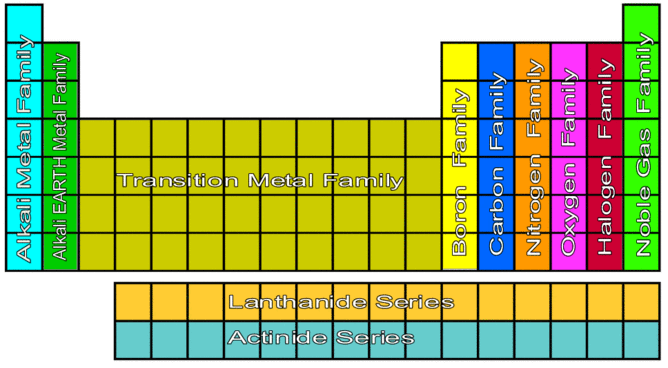
#ALKALI METALS ON PERIODIC TABLE SERIES#
The alkaline metals are a series of elements comprising Group 1 (IUPAC style) of the periodic table: Lithium ( Li), sodium ( Na), potassium ( K), rubidium ( Rb), cesium ( Cs), and francium ( Fr).
